From cuff to cloud: the real-world impact of connected arterial assessment
Peripheral arterial disease remains (PAD) underdiagnosed. Here, we explore how connected solutions like the MESI mTABLET ABI and TBI are streamlining PAD detection from primary care clinics to national screening programs.
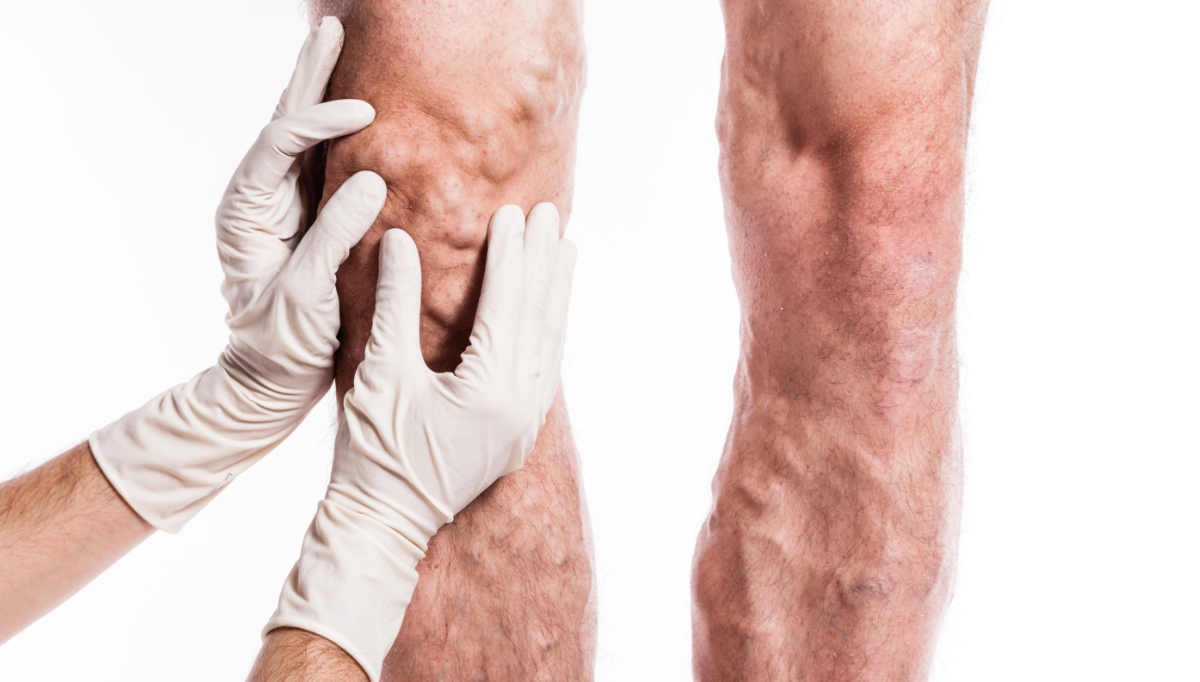
A growing but overlooked disease
The global prevalence of peripheral arterial disease is increasing at an alarming rate. In 2019, there were an estimated 113 million cases of PAD – an increase of 72.5% from 1990 when there were only 65 million individuals affected with this, often silent, disease [1]. Some epidemiological studies suggest even higher figures (236.62 million cases reported in 2015 [2]. Furthermore, projections indicate that by 2050, the number of individuals with PAD could reach at least 360 million, accompanied by a doubling of age-standardized mortality and a dramatic rise in disability-adjusted life years or DALY for short (from 19.7 to 33.1 per 100,000, compared to 2021 data) [3].
This troubling trend is largely driven by the increasing prevalence of metabolic diseases, particularly diabetes [3]. Diabetic individuals are 2 to 3 times more likely to develop PAD than non-diabetics, and nearly 50% of patients with diabetic foot ulcers also suffer from PAD [4], [5], [6]. This comorbidity significantly increases morbidity and premature mortality and is associated with an elevated risk of critical limb ischemia (CLI), the most severe form of PAD with prevalence rates (in diabetic patients) reaching as high as 76% [7]. Diabetics with CLI also suffer worse outcomes (higher rates of lower-extremity amputations and mortality) than those individuals with just one condition. They are at a 5 to 15 times greater risk of major amputation compared to those with PAD alone – CLI itself is associated with amputation rates of 10-40% within six months of diagnosis and mortality rates of 20% in the same period, rising to 50% within five years [8], [9], [10], [11], [12].
Typically, CLI develops gradually and is often preceded by intermittent claudication, a symptom of early-stage PAD. However, only 10% of PAD patients exhibit this symptom, while 40% remain asymptomatic and 50% present with atypical symptoms that may be misattributed to other conditions [13], [14]. Although diabetics are at a higher risk of intermittent claudication (males are 3.5-times more at risk, while females are 8.6-times more at risk than non-diabetics), this has limited diagnostic utility in regard to PAD, as diabetic patients they are already subject to closer medical scrutiny and more likely to receive timely PAD diagnoses [15].
Another problem with intermittent claudication is its tendency to be mistaken for common age-related conditions such as arthritis. Both arthritis and PAD share several risk factors, like advanced age and obesity, which can delay accurate diagnosis and treatment [16], [17], [18]. In addition to arthritis, intermittent claudication may also be confused with other conditions such as sciatica, atheroembolic disease, venous insufficiency, and vasculitis [19]. Lastly, despite increasing efforts to raise awareness about PAD and its symptoms, public understanding remains limited [20], [21], [22], [23]. Many individuals are only diagnosed once the disease has progressed to more advanced stages, underscoring the need for earlier recognition and intervention.
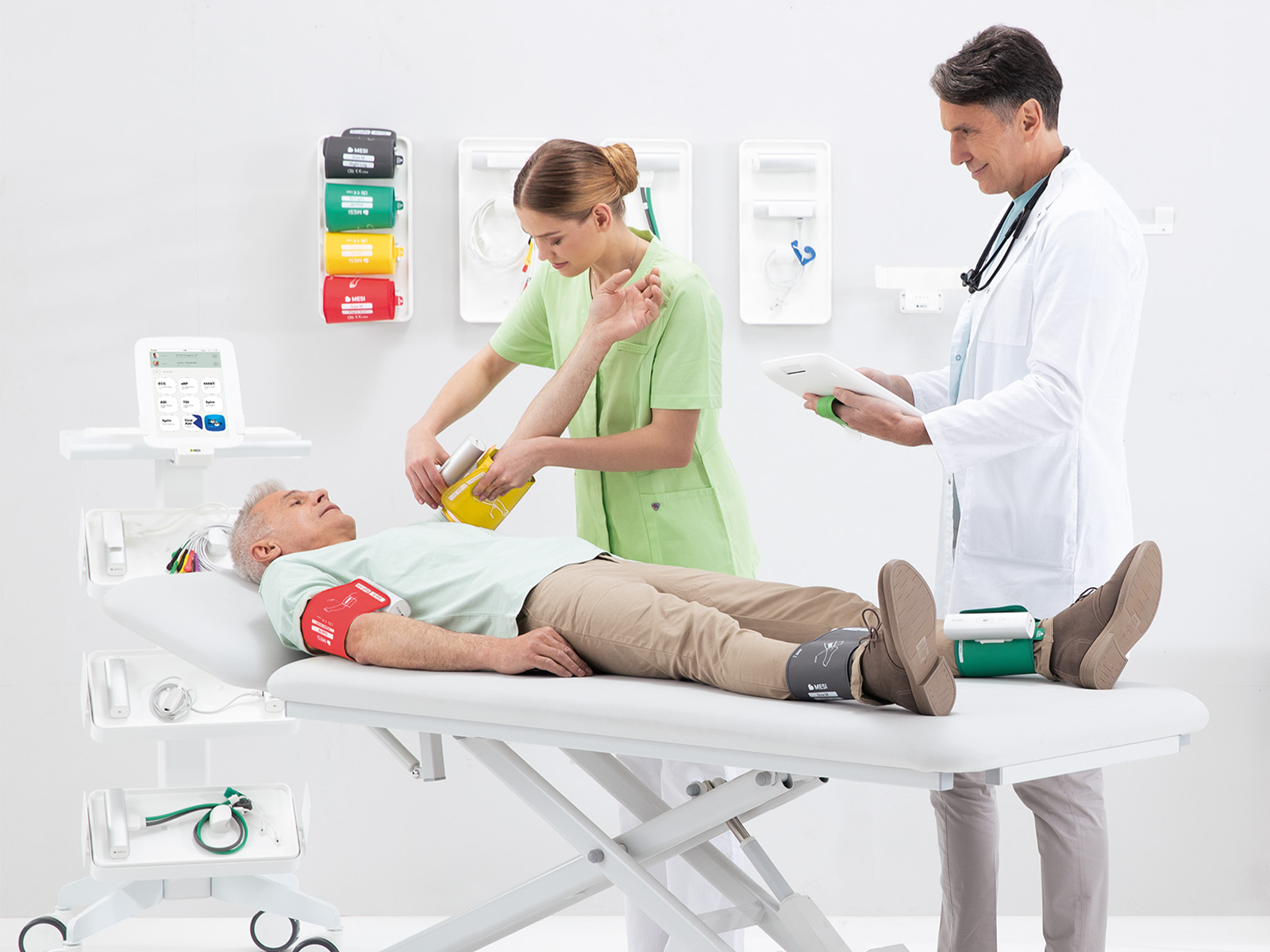
ABI & TBI: simple tools with powerful insights
From a diagnostic standpoint, the ankle-brachial index (ABI) is the superior alternative. Compared to various dubious patient questionnaires or more reliable and accurate but complex imaging techniques like computed tomography angiography (CTA), magnetic resonance angiography (MRA), or ultrasound, ABI is non-invasive, quick, cost-effective, and suitable for outpatient settings [24], [25], [26], [27], [28], [29]. Moreover, beyond diagnosing PAD and assessing its severity, the ABI also has additional diagnostic value.
Research has shown that a low or otherwise abnormal ankle-brachial index (ABI) is a strong indicator of systemic atherosclerosis, not only in the lower extremities but also in coronary and non-coronary (i.e., other than those in the lower extremities) arterial beds [30]. A reduced ABI is also associated with an increased risk of ischemic stroke and the ABI score in general is an important indicator of overall cardiovascular health and can improve the accuracy of cardiovascular risk prediction beyond the FRS (Framingham risk score) [31], [32]. The ABI is also instrumental in diagnosing and guiding treatment of venous and arterial insufficiency ulcers – two most common types of lower-extremity ulcers [33]. They may (visually) look similar, yet they have different pathophysiological mechanisms and require different treatment approaches, which can be mutually exclusive. Fortunately, they can be differentiated using ABI. An ABI between 0.8 and 1.2 indicates venous insufficiency, a lower ABI of between 0.51 and 0.79 suggests mixed venous/arterial ulcers, while a value of 0.5 or lower is consistent with PAD [34].
The ABI is, however, unusable in individuals with incompressible arteries, most caused by medial arterial calcification (also known as Mönckeberg’s sclerosis), which can be found in those with diabetes, renal insufficiency and rheumatoid arthritis [35], [36], [37], [38]. Measuring ABI in these patients is likely to return an abnormally high value (generally in the ≥1.30 range), which suggests higher mortality and greater risk of myocardial infarction than in those with normal ABI, but provides no valuable insight into the presence or severity of PAD [39], [40]. Fortunately, toe arteries are rarely affected by calcification, making them suitable for blood pressure measurement – i.e., toe-brachial index (TBI) assessment – which is performed in a similar fashion to ABI, but with significantly smaller pressure cuffs (for toes) [41]. The TBI is also preferred in individuals with excruciating pain in the lower extremities (due to ulcers or other causes) [42], [43].
Like ABI, TBI provides additional diagnostic insight beyond PAD detection. Low TBI is associated with an increased risk of recurrent cardiovascular disease and progression of diabetic nephropathy in patients with type 2 diabetes; it is also an informative predictor of mortality in dialysis patients [44], [45], [46].
However, when choosing between ABI and TBI assessment for a specific patient, it is important to consider that medial arterial calcification can mask underlying PAD by artificially elevating ABI values into the normal range [47]. Therefore, performing both ABI and TBI is recommended in individuals suspected of having arterial calcification based on their medical history. This approach necessitates access to two separate diagnostic devices – or ideally, a single versatile device with extended functionality that integrates ABI and TBI measurement with an electronic health record (EHR) system, bringing comprehensive PAD assessment into the digital age.
How the MESI mTABLET brings it all together
As a growing body of research shows, comprehensive PAD assessment is no longer an optional procedure but a clinical necessity [48]. Fortunately, with advanced tools like the MESI mTABLET, this process becomes significantly more streamlined compared to traditional methods such as using a Doppler probe and sphygmomanometer. While Doppler-based assessment remains accurate and reliable in the hands of a skilled examiner, it is also time-consuming since it usually takes up to 30 minutes per patient [49].
In contrast, the oscillometric-plethysmographic method, as used by the MESI mTABLET ABI & MESI mTABLET TBI, delivers results in as little as one minute, making it a game-changer for PAD screening [50].

Whether deployed in a local primary care clinic or as part of a nationwide initiative to improve early PAD detection and patient outcomes, the MESI mTABLET offers unmatched ease of use and efficiency. Measuring either the ABI or TBI requires no specialized training beyond knowing where to place the inflatable cuffs – something the tablet itself assists with via an intuitive on-screen guide. Moreover, both ABI and TBI measurements are performed using the same device.
Most importantly, all results obtained using the ABI, TBI, or other diagnostic modules or manually entered in the tablet are automatically stored in the patient’s electronic health record (EHR) via the cloud-based MESI mRECORDS system. This ensures that data is securely saved, easily accessible, and shareable among healthcare professionals using any web-enabled device. It also has many other, EHR-associated benefits like improved data accuracy and reliability due to the elimination of handwriting errors, which reduces malpractice rates [51], [52]. However, the previously stated enhanced accessibility of patient information is the main advantage and paramount in the context of PAD, where effective treatment often requires multidisciplinary collaboration.
PAD patients frequently present with comorbidities such as diabetes, chronic kidney disease, and chronic wounds that interact in complex and mutually reinforcing ways [53], [54], [55]. Due to this, optimal care may involve a vascular surgeon, diabetologist, nephrologist, and wound care specialist – exchanging the patient’s medical data over mRECORDS to formulate a personalized treatment plan with the aim of improving outcome and reducing costs, which can be substantial in managing (advanced) PAD cases [56], [57], [58], [59].
Reducing the burden of PAD therefore rests on two key pillars: timely diagnosis and coordinated, multidisciplinary treatment. The MESI mTABLET, with its speed, ease of use, and smart integration, is uniquely positioned to support both.